Pioneering Proteomic Solutions for Clinical Applications with Mass Spectrometry
Identify and quantify proteins that matter
Mass spectrometry (MS) is the technology of choice for large-scale proteomic monitoring and targeted quantitative bioanalytical analysis.
Sensitive and accurate, it is used for multiple applications, including monitoring of hundreds of targets and precise quantification of individual biomarkers all to support clinical trials and diagnostic development.
CellCarta has led the way in the industrialization of MS platforms by applying them to the needs of the industry.
Our team of scientists has years of experience developing and refining MS workflows for specific applications, all while applying GCLP principles and rigorous QA/QC.
CellCarta has proven expertise working with commercial partners for the development of mass spectrometry-based diagnostics, which led to commercialized diagnostic tests.
Leaders in clinical applications of mass spectrometry
Our scientists continuously strive to develop new ways to harness the power of mass spectrometry. Through their deep understanding of the science, they find solutions to optimize assays based on your needs.
Our proteomic services are driven by the liquid chromatography coupled with multiple reaction monitoring (MRM) platform, which is the backbone for many key applications in all fields – for all phases of clinical trials.
As a targeted platform, MRM provides detailed and quantitative insights into your biomarkers of interest.
Adding to our technical know-how, our proven methods of sample processing, data acquisition and bioinformatics result in scalable workflows generating reproducible results.
We have developed a unique expertise in allowing for the enrichment of the proteome of interest, a key requirement to ensure optimal sensitivity.
We can enrich low-abundance proteins from tissues or relevant biological matrices, including proteins with post-translational modifications.
Analysis can be performed on any species or biological matrices. Workflows for plasma, serum, cells, fresh, frozen, and FFPE tissues, urine, cerebrospinal fluid, sputum, as well as vitreous and aqueous humor are available.
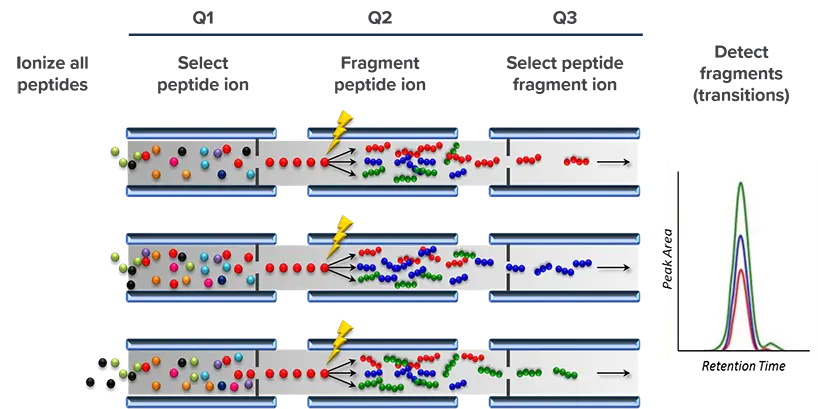
Targeted Platform: Multiple Reaction Monitoring (MRM)
Use of MRM for the analysis of proteins in clinical samples has been increasing due to its high sensitivity and specificity, and its ability to provide absolute quantification.
Its multiplexing capabilities have potential time and cost savings advantages over antibody-based methods.
- Precise multiplexed relative quantification of up to 350 proteins
as follow up on candidates - Precise and accurate multiplexed absolute quantification of
up to 10 proteins for clinical applications - Off-the shelf multiplexed panels or rapid development of custom assays
- Option to couple with hybrid immunoaffinity assays to enrich
low-abundant proteins - Validated assays for clinical use under GCLP and CAP/CLIA environments
Using triple quadrupole or hybrid triple quadrupole/linear ion trap mass spectrometers coupled to upfront liquid chromatography, the MRM platform offers superior specificity, sensitivity and quantification of analytes of interest.
With its double selection approach (Q1-Q3), which improves signal-to-noise and reduces interference, and the multiple measurements per protein, MRM is one of the most specific assays for protein measurement.
Customizing MRM-based assays
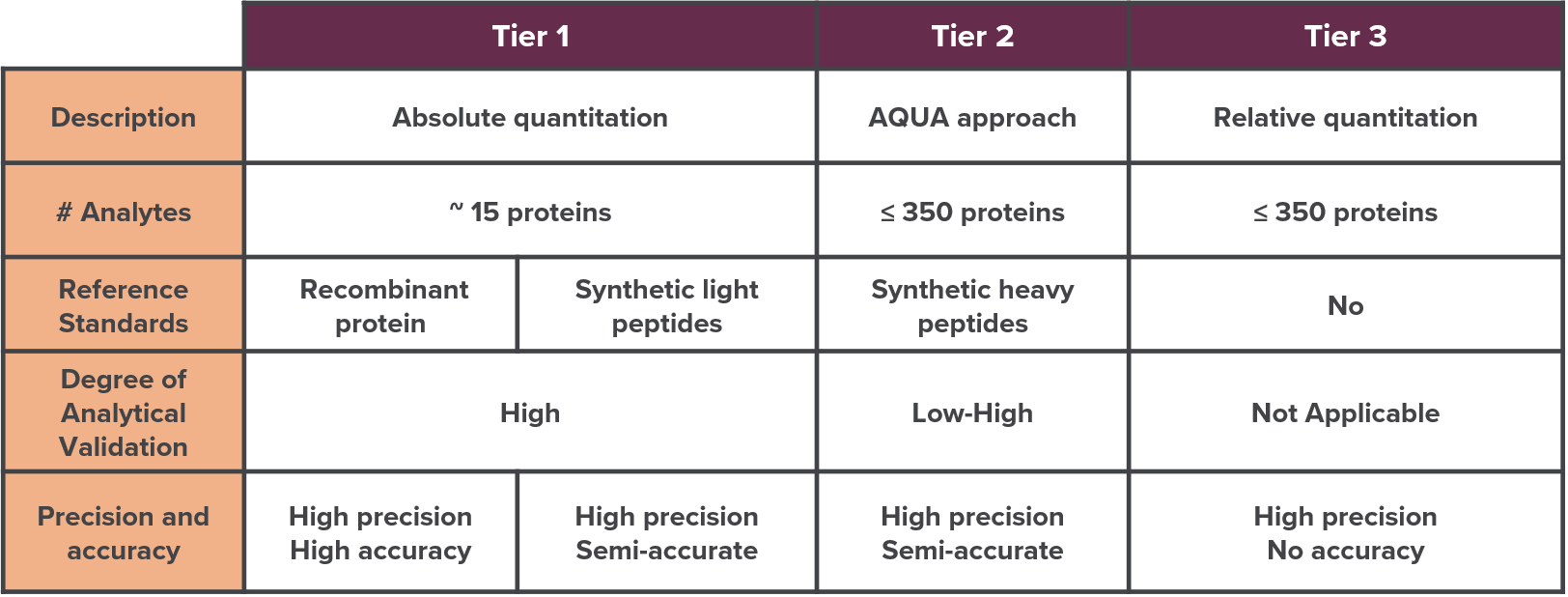
CellCarta offers multiple approaches based on customer needs and study goals.
These range from research-grade assays that measure hundreds of proteins for rapid evaluation of biomarker candidates, to assays that provide absolute quantification of a single or a few proteins for clinical applications and regulatory submission.
MRM: Key Applications
- Monitoring hundreds of targets
- Absolute or relative quantification of individual biomarkers
- Measuring ratios of post-translational modifications