May 8, 2026
Precision medicine depends on the ability to match therapies to the patients most likely to benefit, which in turn relies on diagnostic assays that can accurately detect the biomarkers driving treatment response. For pharmaceutical and biotech developers, these companion diagnostics (CDx) are critical for defining the right patient population and proving therapeutic value in clinical trials, while providing strategic commercial drug product differentiation. Yet, drug-diagnostic co-development is challenging, and creates many practical challenges that can slow programs without well planned coordination.
This is even more the case in China, because of the Chinese regulation “Country Of Origin” which extends the global CDx registration timeline in China. Chinese biotech companies seek China speed with good quality but cannot achieve both easily, while the local IVD companies lacks of a global harmonized lab to meet the China-to-global needs of CDx.
To support the co-development needs of pharma and biotech, CellCarta and Leica Biosystems China, have formed a strategic partnership that brings together their complementary expertise in biomarker assay development and diagnostic manufacturing. The collaboration is designed to provide a smoother, more coordinated and faster path from clinical trial biomarker studies to validated CDx.
THE IMPORTANCE, AND COMPLEXITY, OF CDX DEVELOPMENT
Building a CDx that can withstand clinical and regulatory scrutiny requires tight integration between assay design, validation, and the therapeutic’s own development pathway.
The work done by the laboratory to develop and validate the assays does not automatically transfer when a diagnostic manufacturer steps in. The handoff effectively resets the clock: reagent formulations, scoring algorithms, and validation datasets must be re-established from scratch to meet the manufacturer’s systems and regulatory submission standards. The result is not just additional validation work, it is repeated work, duplicating effort already done, compounding costs, and extending timelines at a stage in development when both are already under pressure.
Therapeutic programs routinely outpace diagnostic validation and manufacturing, opening timing gaps that can stall trial initiation or delay regulatory submission.1 What makes the CellCarta and Leica Biosystems partnership distinct is that collaboration begins well before IUO designation, when assay design decisions still have the greatest leverage. By establishing a shared development framework from the outset, the two organizations carry data forward across every stage rather than regenerating it, following streamlined processes built specifically to bridge laboratory and manufacturing requirements without interruption.
The CellCarta and Leica Biosystems partnership: aligning therapeutic and diagnostic development
CellCarta and Leica Biosystems have partnered to help make CDx development faster and more coordinated. The collaboration combines CellCarta’s expertise in assay development, analytical validation, and regulatory knowledge with Leica’s strength in in vitro diagnostic manufacturing. Together, we offer an end-to-end pathway that connects biomarker assay development with regulatory-ready diagnostics.
How it Works
The two teams follow a parallel workflow and engage each other from the early development of the assay:
- Candidate biomarker assay development:
- Given the breadth of targets under investigation across modalities such as ADCs and TCEs, the partnership allows sponsors to leverage CellCarta’s capabilities to assess a wider biomarker portfolio within a single coordinated framework.
- For those that eventually require CDx, it is transferred to Leica. For those that don’t need CDx, the assay development remains at CellCarta.
- Based on the protocol and intended use, CellCarta can develop and validate exploratory assays or clinical trial assays (CTA) to be used in early-stage clinical trials. In parallel, the existing assay can be transferred to Leica for early kit-based development
- Pivotal trial (Two Scenarios)
- IUO development timeline fits in pivotal trial timeline: Leica develops the investigational-use (IUO) assay based on CellCarta assay that will later form the basis of the final companion diagnostic and will be used in pivotal trial. This is the ideal scenario since no bridging studies are needed.
- IUO development timeline does not fit in pivotal trial timeline: CellCarta develops and validates the clinical trial assay (CTA) to support patient selection for the pivotal trial in China — enabling timely study initiation — with a seamless transition to the Leica IUO assay once available. Both teams share protocols, reagents, and validation data from the outset to maintain technical consistency. A subsequent bridging study compares the CTA and IUO results on patient samples to verify that both assays produce consistent results. The data from the CTA, IUO, and bridging study are then submitted for regulatory review together as a single evidence package.
In either scenario, once the IUO is completed, it is transferred to CellCarta for cross-validation, confirming that the assay performs consistently across both organizations.
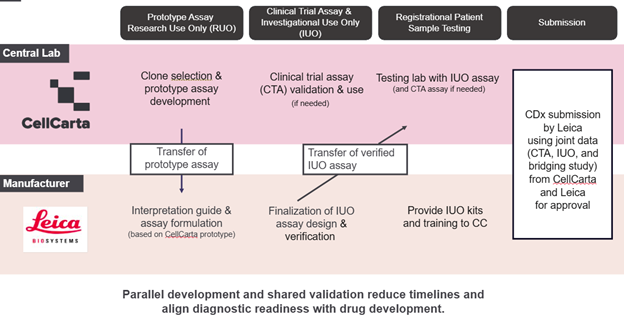
The result is a smoother, more predictable development process that keeps the diagnostic and therapeutic programs moving forward in parallel, helping to prevent trial delays.
What does this mean for sponsors?
The CellCarta–Leica collaboration enables practical advantages that help pharmaceutical and biotech partners manage time, risk, and regulatory complexity in CDx development, offering:
- Navigate Country of Origin restriction: Leica’s manufacturing in China bypasses the Chinese IVD Country of Origin regulation. Global IVD companies without manufacturing facilities in China must comply to this restriction which extends the timeline for China registration.
- Shorter timelines: In the scenario of CTA to IUO bridging in a pivotal trial, CTA validation can be completed within four to five months, allowing patient enrolment to begin while Leica continues IUO development in parallel.
- Lower cost: Through optimized resources and coordinated collaboration—leveraging cost advantages by initiating work in China—this approach reduces costs in the early stages of clinical programs.
- Streamlined process: Joint investment in workflow alignment, including across assay transfer, SOP harmonization, data sharing, and team training, means sponsors inherit a coordinated, ready-to-execute model rather than managing these interdependencies themselves.
- Lower risk: With both the CellCarta CTA and the Leica IUO assay anchored to the same antibody, staining protocol, and scoring algorithm, technical performance is preserved across organizations and across whole stages of clinical trials. This reduces variability and ensures data consistency from trial initiation through to regulatory submission.
- Streamlined submission: With CellCarta’s and Leica’s full commitment to this collaboration and an established data sharing mechanism between the partners, the regulatory submission is very efficient and well-coordinated.
The result is a more predictable development path, which lowers risk and increases the likelihood of clinical trial and CDx development success in China.
Advancing precision medicine through collaboration
China will continue to play an outsized role in oncology and increasingly across other therapeutic areas, particularly in targeted modalities such as T cell engagers and antibody-drug conjugates. These approaches depend on precise biomarker selection, making robust assay strategies and, in many cases, companion diagnostics essential for both clinical development and regulatory success.
Historically, sponsors pursuing companion diagnostic approval in China have faced a binary choice: partner with a local manufacturer, often without a clear global development pathway, or work with a global diagnostics company constrained by Country-of-Origin requirements that can introduce significant delays.
The collaboration between CellCarta and Leica Biosystems establishes a streamlined third pathway for the Chinese market. This integrated model enables sponsors to adopt a China-first, rest-of-world–next strategy while benefiting from coordinated development, regulatory alignment, and cost efficiencies driven by deep, pre-integrated collaboration between the two organizations.
Want to find out more about how this model could support your next therapeutic program? Contact a member of our team.
References
- https://pmc.ncbi.nlm.nih.gov/articles/PMC11835765/
You might also be interested by
Posters
High quality whole transcriptome RNA-sequencing from challenging clinical samples
April 21, 2026
More info