April 14, 2026
External milestones place significant pressure on early-stage and translational programs.
That was the case during a rheumatoid arthritis study conducted for a biotech company at CellCarta’s Lake Forest, California laboratory. The project required the rapid development of two new multiplex immunofluorescence (mIF) assays and their transition into clinical sample testing within a compressed timeline tied to an upcoming conference submission deadline.
Here, we highlight how the Lake Forest team combined rapid assay validation with coordinated clinical testing to deliver ahead of the sponsor’s deadline (Figure 1).
87 samples – both 6-plex mIF assays completed in 6 weeks and 9,000 data points delivered ahead of schedule.
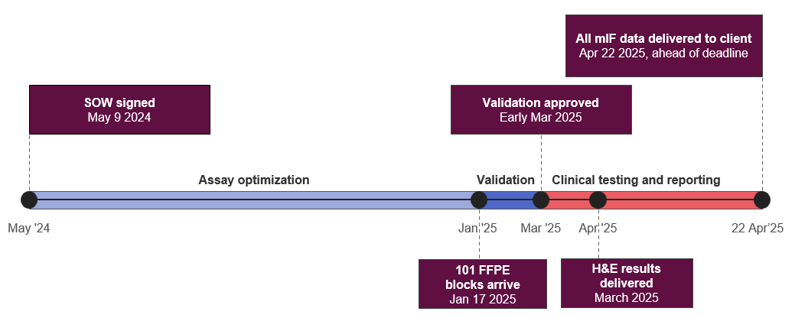
Figure 1: Key milestones of the project at the Lake Forest laboratory. With coordination across teams, the lab was able to move from contract signing to conference-ready data in under 12 months, delivering to the client ahead of schedule.
Optimizing Two New mIF Assays for Rheumatoid Arthritis
The initial phase of the project focused on optimizing two six-plex multiplex immunofluorescence assays on the Akoya platform for use in rheumatoid arthritis tissue samples, a new indication for the Lake Forest Laboratory.
Optimization was carried out between May and December 2024, followed by validation staining and digital image analysis algorithm development in January and February 2025.
Due to the indication, algorithm development proved to be particularly complex, but through careful prioritization and close collaboration between the sponsor, pathology, image analysis scientists, and assay development scientists, the team managed to make quick progress to meet the required deadline.
During this phase, the team also created custom Hematoxylin and Eosin (H&E) evaluation forms for rheumatoid arthritis. These forms were developed to support both assay validation and future clinical sample review, enabling the same tailored pathology evaluation process to be applied across both stages of the project.
Coordinating Development and Clinical Execution
The 101 clinical FFPE block samples arrived at the Lake Forest laboratory on January 17, 2025, while assay validation was being finalized.
To keep the project aligned with the sponsor’s timeline for data delivery by the end of April, the team created a detailed plan for each department to execute as soon as the sponsor approved the full validation package. This close collaboration across assay development, laboratory operations, pathology, digital pathology, and data teams ensured efficient tissue sectioning, staining, and analysis workflows. With this in place, the Lake Forest team was able to:
- Freshly section all FFPE blocks to retain antigenicity, which is critical in mIF testing.
- Deliver H&E results in March, allowing the sponsor to quickly review the samples and select those that would proceed to multiplex immunofluorescence analysis.
- Complete testing and analysis of 87 sponsor-selected samples for both 6-plex mIF assays in just six weeks. All multiplex data, totaling over 9,000 data points (87 samples, each with 105 reported fields), was sent by April 22, ahead of the sponsor’s submission deadline.
- Deliver the results and associated images in ready-to-use formats ahead of the deadline, enabling the sponsor to review the findings and incorporate the data directly into their conference presentation.
Maintaining Momentum When Timelines Are Fixed
Scientific and translational programs often operate against immovable deadlines. When that happens, sponsors need partners who can move quickly without compromising scientific rigor.
At Lake Forest, close coordination between assay development, pathology, and clinical operations allowed complex assay work and large-scale sample testing to proceed in parallel, helping the sponsor deliver critical data in time for their submission milestone.
Looking for a scientific partner for your next study? Connect with our team to find out how we can support your program
About CellCarta’s California, Lake Forest Laboratory
For over 20 years, CellCarta’s CLIA and CAP-accredited Lake Forest laboratory has supported pharmaceutical and biotechnology sponsors across preclinical, discovery, and clinical development programs. Key capabilities include:
- Multiplex immunohistochemistry and immunofluorescence expertise
- Integrated digital pathology and spatial analysis workflows
- In-house tissue biobank access
- Rapid turnaround times designed for early-stage decision-making
Since its opening, the Lake Forest team has worked with over 230 sponsors on over 2,400 unique projects. Operating independently and capable of participating in multi-site joint studies, Lake Forest delivers the speed and flexibility required to advance early-stage programs with confidence.
Find out more about our Lake Forest lab and how it can support your early-stage programs

About the Author:
Andrea Reigel is the Director of US Project Management at CellCarta, leading teams across Lake Forest, CA and Naperville, IL in the delivery of complex clinical trial projects. With a B.S. in Biology, an MBA, and a PMP certification, she brings a mix of scientific knowledge, project management expertise, and strategic leadership to every engagement. At CellCarta, she guides major client partnerships, builds and develops operational teams, and drives process improvements that elevate customer service and project execution.
You might also be interested by
CellTalk Blog
Rescuing a Pilot Study: How a Coordinated Response From CellCarta Kept a Critical Program on Track
April 14, 2026
More infoCellTalk Blog
Emerging Biomarkers Shaping the Next Generation of ADC Therapies
April 14, 2026
More info